Recent research suggests that the gut microbiome, systemic inflammation, and cellular metabolism may influence ovarian aging and the timing of the onset of menopause.
By Ehab Soltan
HoyLunes — Imagine a biological clock. One that not only sets the pace of fertility but also orchestrates the overall vitality of the female body. For centuries, medicine has assumed that this clock has a unique location and limited wind-up, etched in stone from birth. However, cutting-edge science is beginning to hear a different tick, an echo emanating from unexpected places. A new hypothesis suggests that a woman’s reproductive destiny is not written solely in her ovaries; it is the result of a complex, systemic dialogue.
This is no minor debate. Every year, millions of women navigate the menopausal transition, a biological process that can span more than a decade and impact cardiovascular, metabolic, and neurological health. Understanding what truly regulates this chronometer has become one of the central questions in the medicine of aging.
The First Organ to Capitulate to Time
There is an unsettling biological reality that medicine has coolly described for decades: the ovaries are one of the first organs in the human body to age. Before the first wrinkles appear or cardiovascular function shows signs of fatigue, ovarian tissue is already immersed in a process of decline. ([1])
We are born, we are told, with a finite number of follicles. At birth, the ovaries contain approximately one to two million primordial follicles; however, only a small fraction will ever reach ovulation. The vast majority are lost through a process of cellular degeneration known as follicular atresia, a phenomenon that begins even before puberty.
This countdown has been considered inevitable: the exhaustion of the follicular reserve not only marks the end of the reproductive stage but is also the trigger for a cascade of systemic hormonal changes. But if this reserve were the sole determining factor, why does menopause occur at such widely varying ages? Why does the fertility of some women defy statistics while that of others fades prematurely? These questions have opened a rift in the traditional paradigm, driving a transformative quest: What actually calibrates the ovary’s biological clock?
The Cracks in the Classical Paradigm
The dominant model has, for a long time, been linear. Ovarian aging was attributed almost exclusively to the loss of follicles, the deterioration of oocytes, and changes in the hypothalamic-pituitary-ovarian axis. It is a narrative of attrition that explains why fertility decreases after age 35 but ignores the massive influence of external factors.
Today, medicine accepts that the ovary is not a biological island. Population studies demonstrate that nutrition, obesity, chronic stress, and exposure to environmental pollutants can drastically alter the timing of menopause. These findings suggest that reproductive aging responds to far broader systemic influences than previously thought.
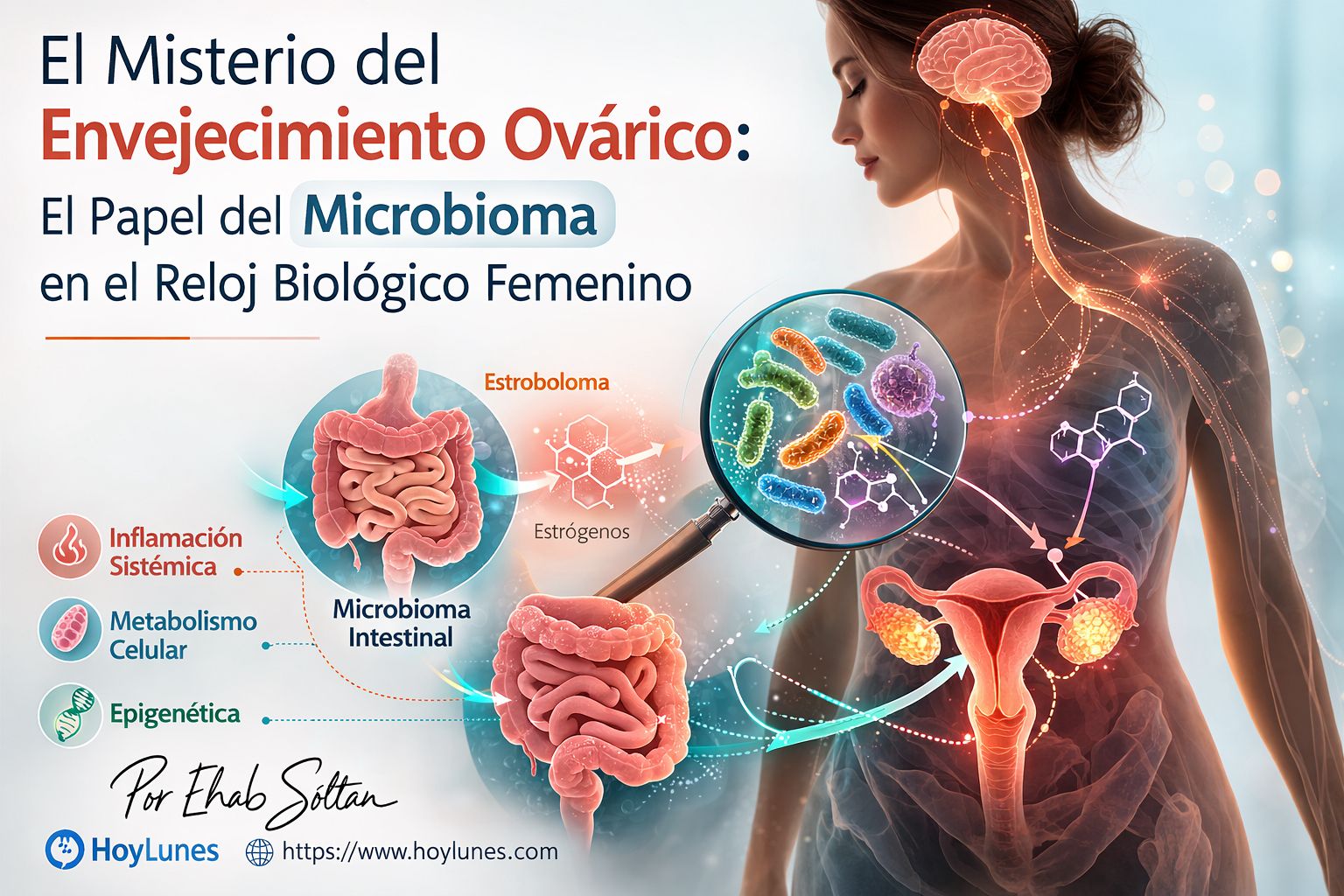
The Microbiome-Ovary Axis: The Hidden Regulator
An unexpected player enters the stage here: the gut microbiome. A bold line of research proposes that ovarian aging is regulated by the trillions of microorganisms that inhabit our intestines. This ecosystem does not just digest food; it constantly interacts with our immune and endocrine systems.
Scientists have identified a specific system termed the estrobolome: a collection of gut bacteria capable of metabolizing estrogens using enzymes such as β-glucuronidase. ([2]) These reactions determine whether hormones are recycled back into circulation or eliminated, indirectly modifying the hormone levels that reach the ovarian tissue. Thus, the gut has the capacity to modulate systemic inflammation and reproductive signaling.
The Experiment That Reshaped Biology
In 2026, a study published in Nature Aging shook the foundations of reproductive biology. ([3]) Cutting-edge researchers tested a radical idea: Could the gut microbiome directly modify ovarian health?
Using animal models, they performed microbiota transplants between young and old subjects. ([4]) The result was surprising: the transplanted microbiome remodeled the ovarian transcriptome, reducing inflammation at a genetic level and generating signals compatible with a rejuvenation of ovarian function.
While translating these findings to the human body will require rigorous clinical studies, the fundamental implication is this: reproductive aging may not depend solely on the “winding” remaining in the ovarian clock, but rather on the quality of the signals it receives from its biological environment.
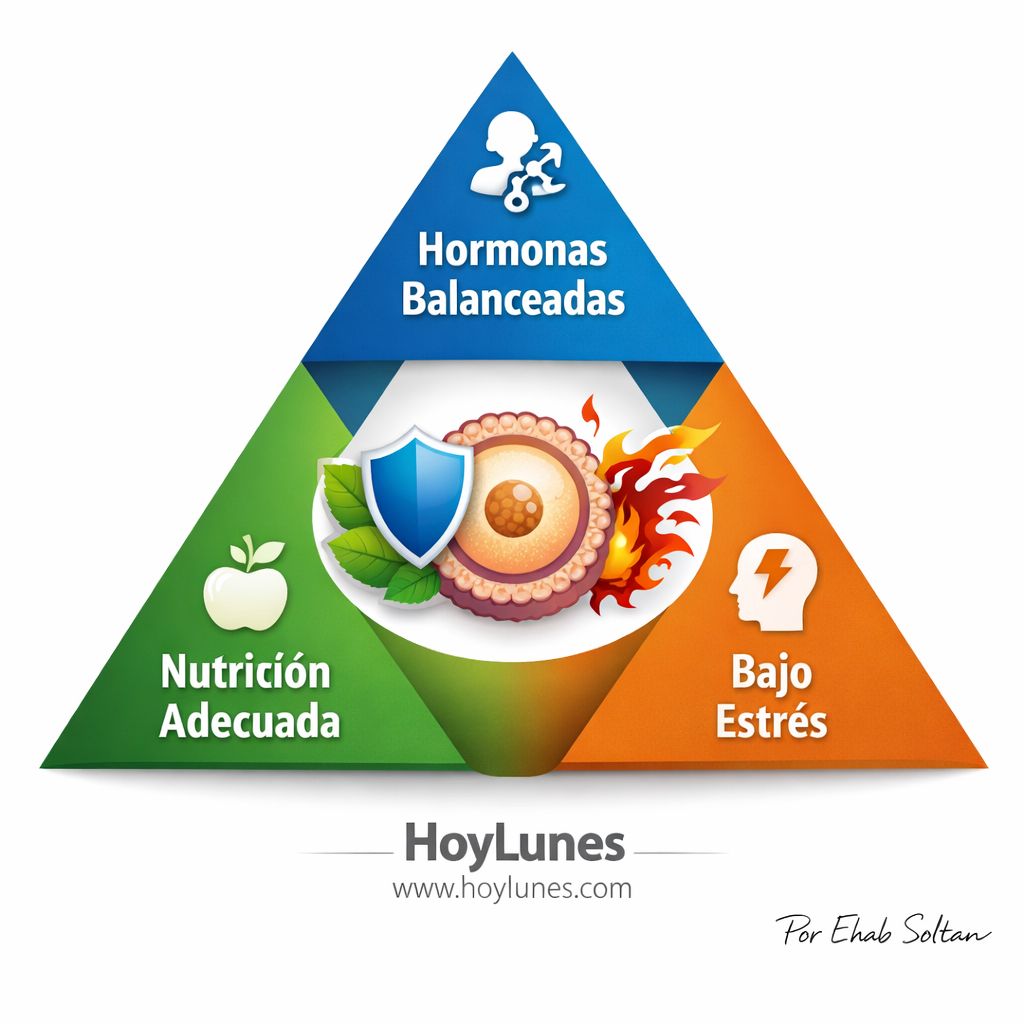
A Triangle of Biological Influences
Current research proposes that ovarian decline is governed by a network where three systems interact: cellular metabolism, epigenetics, and immunological inflammation. ([5])
This metabolic-epigenetic-immunological triangle suggests a vicious cycle: metabolic disturbances affect genetic regulation (epigenetics), these changes alter cellular function, and the resulting inflammation (or inflammaging) accelerates follicular loss. Factors such as oxidative stress, mitochondrial dysfunction, and the accumulation of DNA damage compromise the ability of oocytes to develop normally, acting as accelerators of biological time.
Can We Rewrite Reproductive Destiny?
If the microbiome and metabolism are key pieces, an inevitable question arises: Is it possible to modify the rate of reproductive aging?
This understanding opens doors previously closed. Interventions are already being explored, such as the modulation of the microbiota, the regulation of cellular metabolism, and anti-inflammatory therapies aimed at protecting ovarian tissue. However, the objective is not to prolong fertility indefinitely—which raises considerable ethical challenges—but rather to understand the mechanisms that influence overall women’s health during aging.

Toward a New Medicine for Women’s Health
If this hypothesis is confirmed, menopause will cease to be viewed solely as a “hormonal shutdown” and instead be interpreted as a systemic process. This would imply a new preventive medicine focused not just on hormone replacement but on the care of metabolism, the management of inflammation, and the preservation of microbial ecology. ([6])
The Echo of the Biological Clock
For centuries, we believed that reproductive destiny was written in the ovaries. But today, science suggests that the true biological clock is an invisible network connecting the gut, metabolism, and immunity.
Menopause is not just the end of a reserve; it is the result of an ecosystem that ages together. By understanding this mystery, we do not only rewrite the science of fertility; we also open new avenues for improving women’s vitality throughout their entire lives. Perhaps time is not measured in follicles but in the harmony of our biological network.
Scientific Sources
[1] Study on microbiome and ovarian aging. PubMed. [https://pubmed.ncbi.nlm.nih.gov/38170622/]
[2] Microbiome and female reproductive health. MDPI. [https://www.mdpi.com/3305110](https://www.mdpi.com/3305110)]
[3] Experimental research on microbiota and ovarian function. Nature Aging (2026). [https://www.nature.com/articles/s43587-026-01069-3]
[4] Ovarian transcriptome and microbiota. PubMed. (Referenced in the Nature Aging study).
[5] Metabolic-epigenetic-immunological model of ovarian aging. ScienceDirect. [[https://www.sciencedirect.com/science/article/pii/S2589004226001082]
[6] Scientific review on microbiome and ovarian aging. PubMed. [https://pubmed.ncbi.nlm.nih.gov/38835159/]
This information is for purely informational purposes. To obtain advice or a medical diagnosis, consult a professional.
`#WomensHealth` `#Longevity` `#Microbiome` `#OvarianAging` `#ConsciousMenopause` `#Biotechnology` `#PreventiveMedicine` `#Estrobolome` `#Fertility` `#HolisticWellness` `#EhabSoltan` `#HoyLunes`